The Sequence 10/24-10/30
Insurance-covered Genetic Testing Under-utilized in Patients with Colorectal Cancer, New CRISPR Proteins Discovered, Utility of Polygenic Risk Scores in Predicting Cardiovascular Disease
Insurance-covered genetic testing under-utilized in patients with colorectal cancer
Investigators from Invitae, Optum Labs, UnitedHealthcare, and Variantyx looked at the number of patients with colorectal cancer (CRC) whose insurances covered genetic testing actually received it.
How many patients with colorectal cancer did receive genetic testing?
In a cohort of 55, 595 patients with CRC diagnosed and for whom genetic testing was covered by insurance, only 3.0% (1675 people) actually received genetic testing.
AND, 1 in 6 patients had variants that were clinically actionable.
Woah. Why did so few receive it?
Good question. Most likely, it’s because the insurance policies that universally cover genetic testing for enrollees diagnosed with CRC are fairly new, only taking off in 2020.
In order to understand if the uptake in genetic testing had to do with the newness of this insurance coverage, the group also looked at the uptake of another type of tumor screening in CRC, MSI/IHC (microsatellite instability/immunohistochemistry) tumor screening, since universal coverage of MSI/IHC tumor screening was only implemented in 2009. They found that MSI/IHC was performed in 73.3% (6645 people) of eligible patients during the study period. Although that is much higher than the uptake in genetic testing, it is still disappointing considering how long these policies have been around.
That’s concerning.
It is. As 5% to 15% of CRCs are caused by genetic mutations, or harmful changes in the DNA, genetic testing is an important part of clinical management. Not only is the identification of a genetic cause for CRC informative, it can guide treatment. The patients in this study who had genetic mutations that were clinically actionable were potentially eligible for precision therapy, clinical trials, and/or established management recommendations.
Think you should talk to a genetic counselor about genetic testing? Find one here.
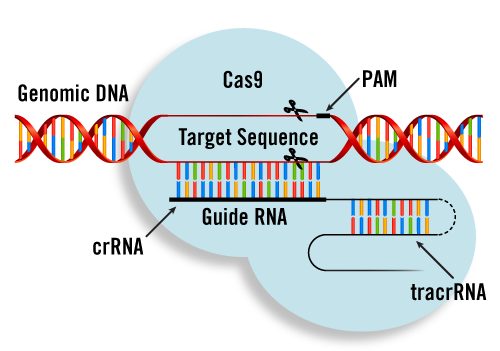
New CRISPR proteins discovered
Urbaitis et al. discovered a new family of CRISPR proteins.
What is CRISPR?
“CRISPR” (pronounced “crisper”) stands for Clustered Regularly Interspaced Short Palindromic Repeats. CRISPR are repeating sequences of genetic code. You have likely heard of them in relation to CRISPR-Cas9 genome editing technology, in which CRISPR can be programmed to target specific stretches of genetic code and to essentially edit DNA at precise locations in the genome.
This is still being studied in the context of genetic conditions, but essentially it would mean being able to edit a genetic mutation, or harmful change in the DNA, curing a genetic disorder caused by a single-gene mutation (think cystic fibrosis, for example).
Photo credit: Mirus Bio LLC
Cool. What’s exciting about a new family of CRISPR proteins?
Firstly, this new CRISPR has the ability to edit in a different area of the DNA than previous CRISPRs. Secondly, the proteins themselves are smaller, making it easier to utilize them in gene therapy. Thirdly, studies have shown they can be used in different organisms.
What’s the takeaway?
I’m an avid believer that gene therapy will be more widely available in my lifetime. It’s discoveries like this that help us find new ways to maneuver the DNA ‘editing’ process in order to make gene therapy a more realistic therapy for genetic conditions.
Credit for this one also goes to a fellow Substack writer for this post. Visit Codon Magazine here:
The utility of polygenic risk scores in predicting cardiovascular disease
A study by Cho et al. examined data on more than 331,000 middle-aged participants in the UK Biobank to evaluate the accuracy of polygenic risk scores for blood pressure in predicting cardiovascular disease.
What is a polygenic risk score?
A polygenic risk score (PRS) is a number, or a ‘score’ that estimates an individual’s risk for a certain condition. They are used in conditions that are caused by changes in many genes, often coupled with environmental factors.
How are they determined?
Let’s use blood pressure as an example, because that is the health outcome studied in this article. Scientists created PRSs for blood pressure by comparing the DNA of patients with different blood pressure levels to determine a ‘collection of genes’ that have more rare variation in the individuals with high blood pressure that are not there in the individuals with lower blood pressure. Then, they can say if you have these ‘X’ number of genes, your PRS for higher blood pressure (i.e. hypertension) is increased.
What did they find?
The group did in fact find that genetically predicted risk for hypertension (i.e. based on a high PRS) was associated with cardiovascular risk. They know this because over time, the risks of cardiovascular disease in patients with normal blood pressure, untreated hypertension, and treated hypertension, respectively, were on par with the risks of cardiovascular disease in patients with genetically predicted rates of normal blood pressure, untreated hypertension, and treated hypertension.
Additionally, they found that based on their PRSs, 1 in 10 individuals with normal blood pressure had comparable cardiovascular disease risks as those with untreated hypertension.
What's the takeaway?
It would be pretty amazing to be able to use a PRS (which is calculated from only a blood sample, by the way) to identify risks for cardiovascular disease; especially in patients with seemingly normal blood pressure. Understanding our risks for common conditions such as cardiovascular disease can guide healthcare management. Hello, precision medicine, it’s nice to meet you.